What Do All Biomolecules Have In Common
Espiral
Apr 03, 2025 · 6 min read
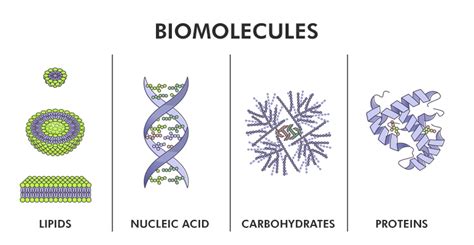
Table of Contents
What Do All Biomolecules Have in Common? Unveiling the Shared Foundations of Life
Life, in all its breathtaking diversity, boils down to a complex interplay of molecules. From the towering redwood to the microscopic bacterium, the fundamental building blocks of life share remarkable similarities. Understanding these commonalities provides crucial insights into the origin, evolution, and functionality of all living organisms. This article delves deep into the shared characteristics of biomolecules, exploring their structural foundations, functional roles, and the interconnectedness that defines the very essence of life.
The Fundamental Building Blocks: Carbon's Central Role
At the heart of every biomolecule lies carbon. This remarkable element, with its four valence electrons, forms the backbone of organic chemistry and is the cornerstone of life's molecular architecture. Carbon's ability to form strong, stable covalent bonds with itself and other elements (like hydrogen, oxygen, nitrogen, and phosphorus) allows for the creation of a vast array of complex and diverse molecules. This versatility is crucial for the formation of the four major classes of biomolecules:
1. Carbohydrates: Energy Sources and Structural Components
Carbohydrates, also known as saccharides, are composed of carbon, hydrogen, and oxygen atoms, typically in a ratio of 1:2:1. They serve primarily as energy sources, with simple sugars like glucose readily metabolized to provide ATP (adenosine triphosphate), the cell's primary energy currency. However, carbohydrates also play vital structural roles. For instance, cellulose forms the rigid cell walls of plants, while chitin provides structural support in the exoskeletons of insects and crustaceans. All carbohydrates are ultimately built from monosaccharide units, the simplest form of sugar, which can link together to form disaccharides (like sucrose) and polysaccharides (like starch and glycogen).
2. Lipids: Diverse Roles in Structure and Function
Lipids are a diverse group of hydrophobic (water-repelling) biomolecules, including fats, oils, waxes, and steroids. While they don't share a common monomeric unit like carbohydrates, they are all characterized by their high proportion of carbon and hydrogen atoms, resulting in their insolubility in water. Lipids play essential roles in:
- Energy storage: Fats and oils store large amounts of energy, providing a long-term energy reserve for organisms.
- Structural components: Phospholipids form the crucial lipid bilayer of cell membranes, regulating the passage of substances into and out of the cell.
- Hormonal signaling: Steroid hormones, such as testosterone and estrogen, are crucial for regulating various physiological processes.
Despite their diversity, lipids all share the defining characteristic of being largely nonpolar, driving their hydrophobic interactions and unique biological functions.
3. Proteins: The Workhorses of the Cell
Proteins are arguably the most versatile class of biomolecules, performing a staggering array of functions within cells. They are polymers composed of amino acids, linked together by peptide bonds to form polypeptide chains. Each amino acid possesses a central carbon atom bonded to an amino group (-NH2), a carboxyl group (-COOH), a hydrogen atom, and a unique side chain (R group). The diversity of R groups dictates the unique properties of each amino acid and, consequently, the overall structure and function of the protein.
Proteins are involved in virtually every cellular process, including:
- Catalysis: Enzymes, which are protein catalysts, accelerate biochemical reactions essential for life.
- Structural support: Collagen and keratin provide structural support in connective tissues and skin, respectively.
- Transport: Hemoglobin transports oxygen throughout the body.
- Defense: Antibodies protect against pathogens.
- Movement: Actin and myosin are involved in muscle contraction.
The remarkable functional diversity of proteins stems from their intricate three-dimensional structures, which are determined by the sequence of amino acids and influenced by various interactions between amino acid side chains.
4. Nucleic Acids: The Blueprint of Life
Nucleic acids, DNA (deoxyribonucleic acid) and RNA (ribonucleic acid), are responsible for storing and transmitting genetic information. They are polymers composed of nucleotide monomers, each consisting of a sugar (deoxyribose in DNA, ribose in RNA), a phosphate group, and a nitrogenous base (adenine, guanine, cytosine, and thymine in DNA; adenine, guanine, cytosine, and uracil in RNA). The sequence of nitrogenous bases in DNA encodes the genetic instructions for building and maintaining an organism. RNA plays crucial roles in translating this genetic information into proteins.
Shared Characteristics Beyond Elemental Composition
While the presence of carbon and the specific elemental compositions are crucial commonalities, the shared characteristics extend far beyond the mere chemical makeup of biomolecules. Several fundamental principles underpin their structure, function, and interaction:
Chirality and Stereoisomerism
Many biomolecules exhibit chirality, meaning they exist as enantiomers—non-superimposable mirror images. This property is particularly important in the context of enzyme-substrate interactions, as enzymes often exhibit high specificity for a particular enantiomer. The selective recognition of specific chiral forms plays a crucial role in biological processes.
Polymerization: Building Complexity from Simple Units
Three of the four major biomolecule classes (carbohydrates, proteins, and nucleic acids) are polymers formed by the joining of smaller monomeric subunits. This process, called polymerization, is facilitated by dehydration reactions, where a water molecule is removed during the formation of each covalent bond between monomers. This fundamental mechanism allows for the construction of incredibly complex macromolecules from simpler building blocks.
Non-Covalent Interactions: Shaping Structure and Function
While covalent bonds form the backbone of biomolecules, non-covalent interactions, such as hydrogen bonds, van der Waals forces, and hydrophobic interactions, play crucial roles in determining their three-dimensional structures and functions. These weaker interactions are responsible for the precise folding of proteins, the formation of DNA's double helix, and many other essential biological processes. The dynamic nature of these non-covalent interactions allows for conformational changes and flexible interactions essential for biological function.
Self-Assembly and Organization
Many biomolecules exhibit a remarkable ability to self-assemble into complex structures. This spontaneous organization, driven by non-covalent interactions and hydrophobic effects, is essential for the formation of cellular structures like membranes, organelles, and even entire cells. The intricate self-assembly processes highlight the inherent order and complexity embedded within the basic building blocks of life.
Dynamic Equilibrium and Regulation
Biomolecules exist in a state of dynamic equilibrium, constantly being synthesized, degraded, and modified within the cell. This dynamic nature allows for cellular adaptation and response to changing environmental conditions. Precise regulatory mechanisms control the synthesis, degradation, and modification of biomolecules, ensuring proper cellular function and homeostasis. This regulatory control is vital for maintaining life's delicate balance.
Interconnectedness: The Web of Life
The four classes of biomolecules are not isolated entities; they are intricately interconnected and interdependent. Carbohydrates provide the energy needed for protein synthesis and the maintenance of cellular structures. Lipids form the structural foundation of cell membranes, while proteins regulate the transport of lipids and other molecules across these membranes. Nucleic acids encode the instructions for building and regulating all other biomolecules. This interconnected network is crucial for the function and survival of all living organisms.
Conclusion: The Unity in Diversity
In conclusion, despite the seemingly limitless diversity of life, all biomolecules share fundamental characteristics rooted in the unique properties of carbon and the principles of molecular assembly and interaction. The commonality in their fundamental building blocks, the shared mechanisms of polymerization, and the reliance on non-covalent interactions underline the underlying unity of life. Understanding these shared features provides a foundation for appreciating the incredible complexity and beauty of the living world and offers invaluable insights into the processes that sustain life on Earth. Further research into the intricacies of these shared characteristics will continue to unravel the secrets of life itself, leading to advancements in diverse fields ranging from medicine and biotechnology to environmental science and understanding the origins of life.
Latest Posts
Latest Posts
-
What Ship Delivered The Atomic Bomb
Apr 04, 2025
-
Gulf Of California On A Map
Apr 04, 2025
-
How High Is The Hoop In The Nba
Apr 04, 2025
-
What Poem Started The Harlem Renaissance
Apr 04, 2025
-
What Size Is A Balance Beam
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Do All Biomolecules Have In Common . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
