Why Does A Salt Solution Conduct Electricity
Espiral
Apr 04, 2025 · 5 min read
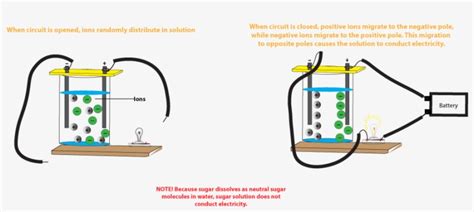
Table of Contents
Why Does a Salt Solution Conduct Electricity? Unveiling the Secrets of Electrolytes
Have you ever wondered why you shouldn't use electrical appliances near water, especially if salt is involved? The reason lies in the fascinating world of electrolytes and their ability to conduct electricity. This article dives deep into the science behind why a salt solution conducts electricity, exploring the concepts of ions, dissociation, and the role of conductivity in various applications.
Understanding Electrical Conductivity
Before delving into the specifics of salt solutions, let's establish a foundational understanding of electrical conductivity. Electrical conductivity is a material's ability to allow the flow of electric charge. Materials are broadly classified into two categories based on their conductivity:
-
Conductors: Materials that readily allow the flow of electric current. Metals like copper and silver are excellent examples. Their conductive properties stem from the presence of freely moving electrons in their atomic structure.
-
Insulators: Materials that strongly resist the flow of electric current. Rubber, wood, and plastics are good insulators because their electrons are tightly bound to their atoms.
The Role of Ions: The Key Players in Conductivity
Unlike metals, the conductivity of salt solutions isn't due to freely moving electrons. Instead, it's attributed to the presence of ions, which are electrically charged atoms or molecules. These ions carry electric charge through the solution. The key to understanding this lies in the process of dissociation.
Dissociation: Breaking Down Salt into Ions
When table salt (sodium chloride, NaCl) dissolves in water, it undergoes dissociation. The strong electrostatic forces holding the sodium (Na⁺) and chloride (Cl⁻) ions together in the crystalline structure are overcome by the polar nature of water molecules. Water molecules surround the ions, effectively separating them and allowing them to move independently within the solution. This process can be represented by the following equation:
NaCl(s) → Na⁺(aq) + Cl⁻(aq)
Where:
- NaCl(s) represents solid sodium chloride.
- Na⁺(aq) represents a sodium ion in aqueous solution.
- Cl⁻(aq) represents a chloride ion in aqueous solution.
This dissociation creates a significant number of mobile charge carriers – the positive sodium ions and the negative chloride ions. These ions are free to move under the influence of an electric field, enabling the solution to conduct electricity.
The Mechanism of Conduction in Salt Solutions
When electrodes (e.g., connected to a battery) are immersed in a salt solution, an electric field is established. This electric field exerts a force on the mobile ions. The positive sodium ions (cations) are attracted to the negatively charged electrode (cathode), while the negative chloride ions (anions) are attracted to the positively charged electrode (anode). This movement of ions constitutes an electric current, allowing the solution to conduct electricity.
Factors Affecting Conductivity of Salt Solutions
Several factors influence the conductivity of a salt solution:
1. Concentration of the Salt Solution
The concentration of the salt solution directly affects its conductivity. A higher concentration means more ions are present in the solution, leading to a greater ability to conduct electricity. A more concentrated solution offers more charge carriers to move under the influence of an electric field. This relationship is not always linear, however, as at extremely high concentrations, ion-ion interactions can impede movement and reduce conductivity.
2. Type of Salt
Different salts dissociate to varying extents. Some salts completely dissociate in water (strong electrolytes), while others only partially dissociate (weak electrolytes). Strong electrolytes, like NaCl, produce a higher concentration of ions and thus exhibit higher conductivity than weak electrolytes. The nature of the ions themselves also plays a role. Larger ions, due to their size and hydration shells, tend to move more slowly than smaller ions, affecting the overall conductivity.
3. Temperature
Temperature plays a significant role in conductivity. Increasing the temperature increases the kinetic energy of the ions. This results in faster movement of ions, hence increased conductivity. However, it is important to note that this relationship is complex and depends on several other factors, such as the solvent's properties and the nature of the ions.
4. Solvent Properties
The solvent used also influences conductivity. Water is a highly polar solvent, effectively dissolving many salts and promoting their dissociation. However, other solvents with different polarities will exhibit varying degrees of effectiveness in dissolving salts, directly impacting the number of ions available for conduction.
Applications of Electrolyte Conductivity
The conductivity of salt solutions has numerous applications in various fields:
1. Electroplating
Electroplating uses the principle of electrolytic conduction to deposit a thin layer of metal onto a surface. A salt solution containing the desired metal ions serves as the electrolyte. The passage of electric current through the solution causes the metal ions to deposit onto the object being plated.
2. Batteries
Batteries utilize electrolytic solutions to facilitate the flow of ions between the electrodes, enabling energy storage and release. The electrolyte in a battery allows the movement of charged ions, completing the electrical circuit and enabling the battery to function.
3. Medical Applications
Electrolyte balance in the human body is crucial for proper physiological function. Analyzing the conductivity of bodily fluids helps diagnose electrolyte imbalances, critical for managing various medical conditions.
4. Water Treatment
Measuring the conductivity of water is an important indicator of its purity and the presence of dissolved salts and minerals. Conductivity measurements are widely used in water treatment to monitor the effectiveness of purification processes.
5. Industrial Processes
Various industrial processes rely on the conductivity of salt solutions. For instance, in chemical synthesis, controlling the conductivity of reaction mixtures can be critical for achieving desired results.
Conclusion: A Deep Dive into Ionic Conduction
The ability of a salt solution to conduct electricity is a direct consequence of the dissociation of the salt into its constituent ions. These mobile ions, under the influence of an electric field, carry electric charge, creating an electric current. Various factors, including concentration, temperature, and the nature of the salt and solvent, significantly influence the conductivity of the solution. Understanding this phenomenon is crucial in numerous applications, ranging from electroplating and batteries to medical diagnostics and industrial processes. Further exploration into the complexities of ionic conductivity reveals a fascinating interplay of chemistry and physics, underscoring its importance in our daily lives and technological advancements.
Latest Posts
Latest Posts
-
The Author Of 2 John And 3 John Calls Himself
Apr 05, 2025
-
Who Was Michael Myers Mask Based On
Apr 05, 2025
-
What State Is Next To Virginia
Apr 05, 2025
-
What Is The State Flower Of Oklahoma
Apr 05, 2025
-
What Did Sir Humphrey Gilbert Think Of The Indians
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Why Does A Salt Solution Conduct Electricity . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
